Edwards Lifesciences: A Market Leader with a Bright Future.
- Glenn
- Jul 15, 2023
- 28 min read
Updated: Apr 4
Edwards Lifesciences is a leading medical technology company focused on treating serious heart valve diseases. Best known for its SAPIEN platform, which allows doctors to replace heart valves without open heart surgery, the company combines trusted products, strong relationships with physicians, and continuous innovation with a highly specialized business model. With growing opportunities in its core valve replacement business, expanding treatments for mitral and tricuspid valve disease, and new therapies under development, Edwards Lifesciences aims to strengthen its position in structural heart care while driving long term growth. The question remains: Does this healthcare leader deserve a spot in your portfolio?
This is not a financial advice. I am not a financial advisor and I only do these post in order to do my own analysis and elaborate about my decisions, especially for my copiers and followers. If you consider investing in any of the ideas I present, you should do your own research or contact a professional financial advisor, as all investing comes with a risk of losing money. You are also more than welcome to copy me.
For full disclosure, I should mention that I do not own any shares in Edwards Lifesciences at the time of writing this analysis. If you would like to copy or view my portfolio, you can find instructions on how to do so here. If you want to purchase shares or fractional shares of Edwards Lifesciences, you can do so through eToro. eToro is a highly user-friendly platform that allows you to get started on investing with as little as $50.
The Business
Edwards Lifesciences was founded in 1958 and has grown into one of the world’s leading structural heart companies. Based in California, the company focuses on technologies that treat serious heart valve diseases, especially in patients who are older, frailer, or otherwise poor candidates for open-heart surgery. Its business is centered around structural heart disease and is divided into three main segments: transcatheter aortic valve replacement, transcatheter mitral and tricuspid therapies, and surgical structural heart products. The largest and most important part of the business is transcatheter aortic valve replacement, often referred to as TAVR. This segment treats patients with aortic stenosis, a condition where the main valve that allows blood to leave the heart becomes too narrow. Instead of opening the chest and replacing the valve through surgery, doctors can insert a new valve through a thin tube, often via a blood vessel in the leg, while the heart is still beating. This allows patients to recover much faster and often leave the hospital within a few days. Edwards’ SAPIEN valve system has become one of the leading products in this market and has helped transform how aortic stenosis is treated around the world. Beyond its leading TAVR business, Edwards is expanding into mitral and tricuspid valve therapies, which represent another large long-term growth opportunity. These valves help control blood flow inside the heart, and when they do not close properly, blood can leak in the wrong direction. Edwards offers both repair and replacement solutions in this segment. Products such as the PASCAL system help repair leaking valves by clipping parts of the valve together, while systems like EVOQUE replace the damaged valve entirely. These minimally invasive therapies are especially valuable for patients who are too weak or too ill to undergo traditional open-heart surgery. This segment is important because it broadens Edwards’ opportunity beyond aortic valve disease and deepens its role within structural heart care. The company also maintains a strong surgical structural heart business. This includes tissue heart valves and other products used in traditional open-heart procedures. While less invasive treatments continue to grow, surgery remains the best option for many patients. Products using Edwards’ RESILIA tissue technology are designed to last longer by slowing down calcium build-up, which can otherwise reduce the life of the valve. This part of the business keeps Edwards relevant across the full treatment pathway and allows it to serve patients regardless of whether they need catheter-based or surgical treatment. Edwards Lifesciences’ competitive moat is primarily built on its clinical evidence and physician trust, high switching costs, regulatory and manufacturing barriers, deep specialization in structural heart, and long-standing relationships with hospitals and clinicians. The strongest part of Edwards’ moat is the trust it has built through decades of clinical evidence and real-world outcomes. In heart valve procedures, doctors and hospitals place enormous importance on safety, durability, and predictability because these are life-critical interventions. Edwards has spent more than sixty years building a reputation for reliable products and strong patient outcomes, particularly through its SAPIEN platform, which has become one of the leading solutions in transcatheter aortic valve replacement. This long track record makes physicians more comfortable continuing to use Edwards’ products and creates a significant advantage over newer competitors. Another important moat is the high switching costs. Structural heart procedures require highly trained teams of physicians, nurses, and specialists who become deeply familiar with a specific device system and procedure workflow. Hospitals invest considerable time and resources in training these teams, building internal expertise, and optimizing processes around Edwards’ technologies. Switching to another provider would require retraining staff, changing procedural routines, and accepting the risk of worse short-term outcomes, which makes hospitals less likely to change suppliers. Edwards also benefits from substantial regulatory and manufacturing barriers. Developing heart valve technologies is highly complex and requires years of research, clinical trials, regulatory approvals, and quality control. The manufacturing process itself is difficult, involving advanced engineering and specialized biological materials such as animal tissue. Strict quality standards and regulatory oversight further raise the barriers to entry, making it very difficult for new competitors to replicate Edwards’ capabilities at scale. Its deep specialization in structural heart disease is another key advantage. Unlike more diversified medical technology companies, Edwards is highly focused on one area of healthcare. This allows the company to concentrate its research, product development, sales efforts, and physician education on becoming the leader in structural heart therapies. This focused expertise strengthens its innovation pipeline and reinforces its position as a trusted specialist. Finally, Edwards has built long-standing relationships with hospitals, heart teams, and clinicians around the world. These relationships go beyond simply selling products. The company provides education, procedural support, and ongoing collaboration with physicians, which helps improve outcomes and builds loyalty over time. Together, these advantages create a durable moat that supports Edwards’ leadership position and long-term growth potential.
Management
Bernard J. Zovighian serves as the CEO of Edwards Lifesciences, a role he assumed in 2023 after joining the company in 2015. His appointment reflects continuity within the organization and a strong strategic focus on structural heart innovation, as he had already played a central role in leading some of the company’s most important growth areas before becoming CEO. Bernard J. Zovighian brings extensive experience in global healthcare and medical technology, having previously held leadership roles at Johnson & Johnson, where he developed deep expertise across multiple medical device segments and international markets. This background gave him strong experience in managing complex healthcare businesses, navigating regulated markets, and driving long term innovation within patient focused industries. Before becoming CEO, Bernard J. Zovighian served as Corporate Vice President and later led Edwards Lifesciences’ transcatheter heart valve business, which is the company’s largest and most strategically important segment. In this role, he was deeply involved in expanding the SAPIEN platform, strengthening physician relationships, and supporting the continued global adoption of transcatheter valve therapies. Because this segment represents the majority of the company’s revenue and is at the core of its competitive moat, his experience leading this business provides strong operational continuity and strategic credibility as CEO. Bernard J. Zovighian holds dual master’s degrees in science and business from the Universities of Marseille in France. His academic background reflects a combination of technical understanding and business leadership, which is particularly valuable in a highly specialized medical technology company where innovation, clinical evidence, and commercial execution must work closely together. Since becoming CEO, Bernard J. Zovighian has emphasized Edwards Lifesciences’ patient centered culture and long term innovation strategy. He has repeatedly highlighted the company’s mission of addressing unmet patient needs through breakthrough therapies and expanding access to minimally invasive heart valve treatments. Under his leadership, the company continues to focus on its large untapped patient opportunity, particularly within structural heart disease where many patients remain untreated today. Bernard J. Zovighian has also expressed confidence in the company’s pipeline, especially in mitral and tricuspid therapies, which could become important future growth drivers beyond the core TAVR business. Another point worth adding is Bernard J. Zovighian’s strong emphasis on people and culture. Edwards Lifesciences has long been known for its physician partnerships, employee engagement, and patient first mindset, and Bernard J. Zovighian has made it clear that preserving this culture is a strategic priority. In medical technology, strong culture matters because success depends not only on product innovation but also on close collaboration with physicians, hospital systems, regulators, and internal research teams. His people focused leadership style appears well aligned with this model. Bernard J. Zovighian appears well positioned to lead Edwards Lifesciences through its next phase of growth. His deep experience inside the company, leadership of its most important business segment, and clear focus on long term patient outcomes and innovation suggest that Bernard J. Zovighian is indeed the right person to guide the company forward.
The Numbers
The first number we will look into is the return on invested capital, also known as ROIC. We want to see a 10-year history, with all numbers exceeding 10% in each year. Edwards Lifesciences has historically delivered very strong ROIC, which is exactly what you would hope to see from a high quality medical technology company with a strong competitive moat. Even though ROIC has declined from 23,4% in 2021 to 13,1% in 2025, it still remains comfortably above 10%, which suggests that the company continues to create solid value from the money it uses in the business. The decline does deserve an explanation, but it does not necessarily mean that the business itself has become weaker. Several structural characteristics explain why Edwards Lifesciences has historically generated high ROIC. First, the company benefits from strong profitability because it is a leader in structural heart, especially through its SAPIEN platform in transcatheter aortic valve replacement. These are highly specialized, life saving products where doctors care deeply about safety, reliability, and proven outcomes. Because of this, Edwards Lifesciences can maintain strong margins, which is one of the main reasons ROIC has historically been high. Second, Edwards Lifesciences has been able to generate a lot of profit without needing huge amounts of money tied up in factories, equipment, and other physical assets compared to many industrial companies. Of course, the company still invests in manufacturing, research, and clinical studies, but it does not need massive plants or expensive machinery on the same scale as an industrial manufacturer. This has helped the company produce strong returns for many years. The main reason ROIC has declined since 2021 is likely that Edwards Lifesciences has been investing heavily for future growth. Over the last several years, the company has spent significant resources expanding beyond its core aortic valve business into mitral and tricuspid therapies. These newer areas are growing quickly but are still smaller and not yet as profitable as the main TAVR business. In simple terms, the company is spending money today to build products and market positions that it expects to generate larger profits in the future. That often causes ROIC to decline in the short term. Another reason is that the company has made acquisitions and expanded into adjacent areas, which increases the amount of money tied up in the business immediately, while the profit contribution from those investments usually takes time to show up. This can temporarily lower ROIC even if those investments end up being very successful later on. It is also worth noting that the TAVR business has matured compared to the exceptionally strong growth years around 2020 and 2021. When growth slows a bit while the company continues to invest aggressively in new therapies and innovation, ROIC often comes down naturally. I would therefore not see the decline as a major red flag, but rather as part of the company moving into its next growth phase. Looking ahead, I do expect ROIC to improve over time, although it may not return to the very high levels above 20% that we saw earlier in the decade. The most likely scenario is that ROIC gradually moves higher as the mitral and tricuspid businesses become larger and start contributing more profit. If those newer therapies scale successfully, the money Edwards Lifesciences is investing today should begin to generate stronger returns in the coming years.

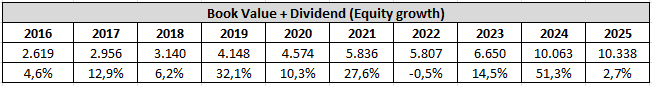
The next numbers are the book value + dividend. In my old format this was known as the equity growth rate. It was the most important of the four growth rates I used to use in my analyses, which is why I will continue to use it moving forward. As you are used to see the numbers in percentage, I have decided to share both the numbers and the percentage growth year over year. To put it simply, equity is the part of the company that belongs to its shareholders – like the portion of a house you truly own after paying off part of the mortgage. Growing equity over time means the company is becoming more valuable for its owners. So, when we track book value plus dividends, we’re essentially looking at how much value is being built for shareholders year after year. Edwards Lifesciences has managed to increase its equity in nine out of the past ten years, which is a very strong and encouraging trend. This tells us that the company has consistently built value for shareholders over time. The main reason for this steady growth is that Edwards Lifesciences has remained highly profitable for many years. Because the company operates in specialized medical devices with strong margins, especially in structural heart, it has been able to generate consistent profits and retain a meaningful portion of those profits in the business. Those retained earnings directly increase equity over time. Another important reason is the company’s long term focus on reinvestment. Edwards Lifesciences has consistently invested in research and development, clinical trials, product innovation, and acquisitions that strengthen its position in structural heart. While these investments can weigh on short term profitability, they help build the long term value of the business and support future earnings growth. Over time, this strengthens the balance sheet and contributes to rising equity. The particularly large increase from 2023 to 2024, where equity rose by 51,3%, stands out and should be viewed separately from the normal operating trend. This was largely driven by the sale of the company’s Critical Care division to BD (Becton, Dickinson and Company), which significantly boosted net income and therefore equity. Because this was a major one time transaction, it is not something we should expect to repeat every year. Looking ahead, I do expect equity to continue increasing over time, although likely at a more normal pace than what we saw in 2024. The main reasons are still in place. Edwards Lifesciences continues to generate strong profits, has a leading position in structural heart, and is investing in growth areas such as mitral and tricuspid therapies. As these newer therapies mature and contribute more meaningfully to earnings, they should support further growth in equity. That said, the pace of growth may be lower in some years because the company is currently investing heavily in future products and innovation. This can temporarily slow equity growth, but if these investments succeed, they should create more shareholder value over the long term.
Finally, we will analyze the free cash flow. Free cash flow, in short, refers to the cash that a company generates after covering its operating expenses and capital expenditures. I use levered free cash flow margin because I believe that margins provide a better understanding of the numbers. Free cash flow yield refers to the amount of free cash flow per share that a company is expected to generate in relation to its market value per share. Edwards Lifesciences has historically generated positive free cash flow every year over the past decade, which is exactly what you would hope to see from a high quality medical technology company. While free cash flow has fluctuated from year to year, it reached its highest level in 2025 at 1.335, which is a very encouraging sign after the weaker levels seen in 2023 and especially 2024. One of the main reasons Edwards Lifesciences has consistently generated strong free cash flow is the nature of its business model. The company operates in specialized medical devices with strong margins, particularly in structural heart therapies such as the SAPIEN platform. These products are highly specialized, have strong pricing power, and benefit from physician trust and clinical evidence, which supports solid profitability. Because of this, a large share of the company’s earnings can eventually be converted into cash. The decline in free cash flow in 2023 and 2024 was mainly driven by temporary factors rather than a weakening of the business. In 2024, the biggest impact came from unusually large tax payments related to the sale of the Critical Care division as well as ongoing tax matters. These were largely one time items that reduced cash generation in that year. In addition, Edwards Lifesciences made acquisitions to strengthen its structural heart business, which also reduced free cash flow in the short term because cash was used upfront to support future growth. There were also operational headwinds such as weather related disruptions, shortages of IV fluids, and pricing adjustments in China, all of which temporarily pressured cash generation. The strong rebound in 2025 suggests that many of these temporary headwinds have faded. Free cash flow not only recovered but reached the highest level in the decade, while the free cash flow margin also rebounded strongly to 22,0%. To me, this is a very positive sign because it shows that the weakness in 2024 was not structural. Looking ahead, I do expect free cash flow to grow over time, although it will likely continue to fluctuate from year to year. The most important driver will be continued growth in the core TAVR business and the scaling of the mitral and tricuspid therapy segments. As these newer therapies become larger and more profitable, they should contribute meaningfully to cash generation. At the same time, Edwards Lifesciences is continuing to invest in production capacity to support demand across TAVR, TMTT, and Surgical, which management has highlighted as its first capital allocation priority. These investments may create some short term fluctuations, but they should support stronger cash generation in the future. Edwards Lifesciences uses its free cash flow in a very disciplined way and management has been clear about its capital allocation priorities. First, the company reinvests in the business. This includes expanding production facilities, increasing capacity, funding research and development, and supporting the launch of new therapies. These investments are especially important as demand continues to grow across TAVR, transcatheter mitral and tricuspid therapies, and the surgical segment. Second, Edwards Lifesciences uses part of its free cash flow for external growth through acquisitions and minority investments, typically smaller deals focused on structural heart. These acquisitions are meant to strengthen the company’s product pipeline, expand its technology platform, and support long term growth. The largest use of free cash flow after reinvestment has been share repurchases. Returning capital to shareholders through buybacks has been an important part of Edwards Lifesciences’ strategy for many years. In 2025 alone, the company repurchased just under $900 million of its own shares, and management still has approximately $2 billion remaining under its authorization. The free cash flow yield is at its highest level in more than a decade. While this does not necessarily suggest that the shares are cheap, it does indicate that Edwards Lifesciences is trading at its most attractive valuation in more than a decade based on free cash flow. However, we will revisit valuation later in the analysis.

Debt
Another important aspect to consider is the company’s level of debt. It is essential to assess whether a business maintains a manageable debt level that can be repaid within three years, which is determined by dividing the total long term debt by earnings. For Edwards Lifesciences, this calculation shows that the company has only 0,57 years of earnings in debt, which is well below the three year threshold. Consequently, debt is not a concern in this case. Historically, Edwards Lifesciences has maintained a low level of debt, reflecting a conservative and disciplined approach to financial management. This track record suggests that debt is unlikely to become a significant issue for the company in the future. Combined with consistent profitability and strong cash generation, the company’s balance sheet remains a source of strength and flexibility. This financial strength also gives Edwards Lifesciences the ability to continue investing in research and development, expand production capacity, pursue acquisitions, and return capital to shareholders through share repurchases without putting pressure on the business.
Support the Blog
I want to keep the blog free and accessible for everyone. If you enjoy the content and would like to support it, you can buy me a cup of coffee through PayPal. Every little bit helps and is truly appreciated!
Risks
Competition is a risk for Edwards Lifesciences because the structural heart market is highly competitive, innovation driven, and constantly evolving. Unlike many industries where products can remain unchanged for long periods, cardiovascular medical technology is characterized by continuous product development, clinical trials, and technological advances. This means that Edwards Lifesciences must consistently improve its therapies to maintain its leadership position. If competitors introduce products with better clinical outcomes, easier procedures, stronger durability, or lower cost, hospitals and physicians may gradually shift toward those alternatives. The most direct competitive pressure comes from large and well established medical technology companies such as Medtronic and Abbott Laboratories. In transcatheter aortic valve replacement, which is Edwards Lifesciences’ most important business segment, Medtronic remains a major competitor with its Evolut platform, while Abbott also competes in several structural heart categories. These companies are large, well funded, and have extensive research capabilities, global sales networks, and strong relationships with hospitals. This means Edwards Lifesciences is not only competing against smaller innovators, but also against some of the strongest players in global medical technology. Competition can affect Edwards Lifesciences in several ways. One risk is product innovation. In structural heart, even small improvements in safety, durability, ease of use, or recovery time can influence physician preference. If a competitor develops a valve system that offers better long term outcomes, fewer complications, or a simpler implantation procedure, doctors may increasingly prefer that solution. Because these products are used in life critical procedures, clinical evidence and physician confidence are extremely important. This means that Edwards Lifesciences must continue investing heavily in research, development, and clinical studies simply to maintain its competitive position. Another important risk is technological disruption. The company itself highlights that present or future products could become less attractive as a result of technological advances by competitors or by alternative therapies, including drug based treatments. While minimally invasive valve procedures are currently a major growth area, future innovations in cardiovascular medicine could change treatment pathways over time. If competing technologies or even pharmaceutical solutions reduce the need for certain procedures, this could affect Edwards Lifesciences’ long term growth. Competition also extends beyond products themselves and includes pricing and value demonstration. Healthcare systems around the world are increasingly focused on cost effectiveness and value based care. Hospitals, payors, and governments want evidence that a therapy not only improves patient outcomes but also reduces overall healthcare costs, such as shorter hospital stays or fewer complications. If competitors can demonstrate similar clinical outcomes at lower cost, Edwards Lifesciences may face pricing pressure, which could impact margins.
Laws and regulations are a risk for Edwards Lifesciences because the company operates in one of the most heavily regulated industries in the world. Nearly every part of its business is closely monitored by governments and health authorities, from product development and clinical testing to manufacturing, labeling, marketing, and post market monitoring. Because Edwards Lifesciences sells high risk medical devices that are used in life critical heart procedures, the regulatory requirements are especially strict. This means that delays, rule changes, or compliance issues can directly affect the company’s ability to launch products, grow revenue, and maintain profitability. One of the biggest regulatory risks comes from product approvals. In the United States, the U.S. Food and Drug Administration plays a central role in deciding whether Edwards Lifesciences can commercialize new products or expand the use of existing ones. For products such as heart valves, the approval process is often long, expensive, and data intensive. The company must provide extensive clinical evidence proving that a product is safe and effective. If the FDA requests additional studies, asks for more data, or delays approval, product launches can be pushed back significantly. This can slow growth and allow competitors more time to strengthen their market position. Even after a product has been approved, regulatory risk does not disappear. Authorities can require additional monitoring, clinical follow up studies, or new safety reporting procedures. If issues arise in real world use, the FDA has the authority to issue warnings, impose fines, request product recalls, suspend sales, or in severe cases withdraw approvals altogether. Because Edwards Lifesciences’ products are used in critical heart procedures, even a small safety issue could have serious consequences both financially and reputationally. Another important risk is the constantly changing regulatory environment outside the United States. In Europe, the Medical Device Regulation has significantly increased the burden on medical device companies by requiring stronger clinical evidence, more documentation, stricter labeling standards, and enhanced post market surveillance. These stricter rules increase both the time and cost required to get products approved and keep them on the market. Delays in obtaining approvals can disrupt product supply and limit Edwards Lifesciences’ ability to compete effectively in important international markets. Regulation can also affect how Edwards Lifesciences interacts with physicians, hospitals, and healthcare systems. Laws such as anti kickback rules, false claims regulations, and transparency requirements around physician payments are particularly important in healthcare. Because Edwards Lifesciences works closely with physicians through training, clinical support, and educational programs, any failure to comply with these rules could lead to investigations, fines, legal costs, and reputational damage.
Reliance on third party suppliers is a risk for Edwards Lifesciences because the company depends on outside vendors for several critical parts of its operations, including raw materials, specialized components, manufacturing support, logistics, and sterilization of medical devices. Since Edwards Lifesciences operates in highly specialized structural heart therapies, any disruption in this supply chain can directly affect its ability to produce and deliver life saving products to hospitals and patients. One of the biggest risks is the company’s reliance on single source suppliers for certain materials and services. In some cases, Edwards Lifesciences uses only one approved supplier because of quality requirements, cost efficiency, availability, or strict regulatory standards. While this helps ensure consistency and product quality, it also creates concentration risk. If one of these suppliers faces operational issues, financial distress, or chooses to stop serving the medical device industry, Edwards Lifesciences may not be able to replace that supplier quickly. This risk is made more serious by the regulatory complexity of the industry. Switching suppliers is not as simple as choosing a new vendor. Any new supplier often needs to go through a lengthy approval and validation process to meet the standards of regulators such as the U.S. Food and Drug Administration and other health authorities worldwide. This means that even if an alternative supplier exists, it can take significant time and effort to qualify that supplier and integrate it into the production process. During that time, product availability could be affected. The sterilization process is another critical point of dependency. Medical devices such as heart valves must meet extremely strict safety standards before they can be used in patients. If a third party sterilization partner experiences delays, equipment failures, quality issues, or regulatory problems, this could slow shipments and disrupt product supply. In an industry where timely access to life saving technologies is essential, even short delays can have meaningful consequences. Another important point is demand scaling. As Edwards Lifesciences continues to grow its TAVR, transcatheter mitral and tricuspid therapies, and surgical businesses, production capacity must keep pace with demand. If suppliers are unable to scale quickly enough, the company may face shortages just as demand is increasing. This could lead to missed sales opportunities and allow competitors to gain share. Finally, any supplier related issue can also become a reputational risk. If quality problems lead to recalls, safety alerts, or delays in procedures, hospitals and physicians may lose confidence in the company’s products. In medical technology, trust and reliability are extremely important, which means supplier issues can have consequences that extend beyond short term financial results.
Reasons to invest
The transcatheter aortic valve replacement (TAVR) segment is a reason to invest in Edwards Lifesciences because it remains the company’s largest business, benefits from strong clinical evidence, and still offers a long runway for durable growth. TAVR is built around the SAPIEN platform, which has become one of the most trusted and widely used therapies for severe aortic stenosis. What makes this segment especially attractive is that it combines current scale with continued expansion opportunities, which is a powerful combination for long term investors. One of the most important reasons to be positive on the TAVR segment is the strength of the clinical data supporting the SAPIEN platform. Long term results from the PARTNER II and PARTNER III studies have reinforced physician confidence in the durability and performance of Edwards’ valves. In healthcare, long term clinical data is extremely valuable because it directly influences physician decisions, treatment guidelines, and reimbursement policies. The seven year and ten year data showing strong durability helps reassure doctors that the valves can perform well over many years, which is especially important as younger patients increasingly become eligible for treatment. Another major growth driver is the EARLY TAVR trial. This study has been particularly important because it supports earlier treatment of severe aortic stenosis rather than waiting for symptoms to worsen. Historically, many patients were monitored under a watchful waiting approach. The new data supports a shift toward more proactive treatment, which can expand the number of patients who receive TAVR and accelerate procedure volumes. This is a meaningful change because it does not just help Edwards Lifesciences take share, it can also expand the total market. The guideline changes in Europe further strengthen this opportunity. Moving away from watchful waiting toward earlier intervention represents one of the most significant shifts in the treatment pathway in many years. While management has emphasized that these changes will not act like a light switch overnight, they do create multiple layers of durable growth as the new standards gradually become embedded in clinical practice. The concept of lifetime management is another reason to invest in the TAVR segment. Edwards Lifesciences has focused heavily on ensuring that patients have options for future procedures, whether that means valve in valve treatments or other follow up interventions later in life. This is especially relevant as patients are treated earlier and potentially live long enough to require a second or even third procedure. By positioning SAPIEN as a platform that supports lifetime management, Edwards Lifesciences strengthens its long term relevance and competitive advantage.
The growing Transcatheter Mitral and Tricuspid Therapies (TMTT) segment is a reason to invest in Edwards Lifesciences because it represents one of the company’s most important long term growth opportunities beyond the core TAVR business. While TAVR remains the largest segment today, TMTT gives Edwards Lifesciences a second major growth engine that can materially expand revenue over the coming years. Management’s expectation of reaching around $2 billion in revenue from this segment by 2030 highlights the scale of the opportunity. One of the most attractive aspects of the TMTT segment is the strength of the product portfolio. Unlike many competitors that may only offer a single solution, Edwards Lifesciences now has both repair and replacement therapies for both mitral and tricuspid valve disease. This includes PASCAL for repair, EVOQUE for tricuspid replacement, and SAPIEN M3 for mitral replacement. Having a full toolbox of therapies is important because patients with mitral and tricuspid regurgitation often present with very different disease characteristics. Some patients are better suited for repair, while others need replacement. By offering multiple solutions, Edwards Lifesciences becomes a stronger strategic partner for physicians rather than simply a single product supplier. The launch of SAPIEN M3 is a particularly important reason to be optimistic about the segment. This product expands Edwards Lifesciences’ mitral offering and represents the company’s first transcatheter replacement option for certain mitral patients in the United States. Because it is still very early in the rollout, the revenue contribution today is modest, but management has expressed strong confidence in its long term potential. Early physician feedback and strong patient outcomes are encouraging signs that this therapy could become a meaningful contributor to future growth. Another reason to invest in this segment is that Edwards Lifesciences is still in the early stages of market penetration. Many of these therapies are only beginning to scale across geographies and hospital centers. This means there is still significant room for center expansion, physician training, and broader adoption over the coming years. As with TAVR in its earlier years, the opportunity is not only about market share but also about expanding the overall treatment market. The strategic importance of this segment also lies in how it complements the company’s broader structural heart leadership. By building a strong presence in mitral and tricuspid therapies, Edwards Lifesciences deepens its relationships with physicians and hospital systems. This strengthens the overall moat and supports cross selling and physician loyalty across the structural heart portfolio.
Innovation and expansion into new therapies are a reason to invest in Edwards Lifesciences because they provide the company with multiple long term growth drivers beyond its already strong core businesses in TAVR and TMTT. While the existing platforms are already large and growing, one of the most attractive parts of the investment case is that Edwards Lifesciences continues to build entirely new therapy areas and launch next generation products that can expand its addressable market over many years. One of the key reasons this matters is that Edwards Lifesciences does not simply rely on its current products. Management has made it clear that every major platform has a next generation version in development. This includes updated versions of important therapies such as PASCAL and EVOQUE, with the next generation PASCAL expected to launch later in 2026. This constant product improvement is important because in medical technology, better outcomes, easier procedures, and improved durability can directly support stronger adoption and market share gains. Another reason innovation is attractive is the company’s expansion into new therapeutic areas that are adjacent to its core expertise. One example is aortic regurgitation, which is different from aortic stenosis and requires a different valve solution. Edwards Lifesciences has been developing this area through its acquisition of JC Medical. Management has highlighted that this condition is significantly undertreated today because many patients only have surgical options available. A successful transcatheter solution here could open an entirely new growth market. The company is also expanding into structural heart failure through technologies such as Cordella, which was added through the acquisition of Endotronix. This device helps physicians monitor patients with heart failure and better manage treatment over time. This is strategically important because it moves Edwards Lifesciences beyond valves alone and deeper into the broader structural heart ecosystem. The company’s innovation pipeline also reflects a strong culture of research and development. Management has explicitly described innovation as the lifeblood of the company. This is important because long term success in medical technology depends heavily on continuously developing better solutions for unmet patient needs. Edwards Lifesciences has shown over decades that it can translate research into commercially successful products. In addition to internal innovation, the company actively invests in external growth through smaller acquisitions, minority investments, and seed funding for early stage companies. This disciplined approach allows Edwards Lifesciences to supplement its internal pipeline with promising technologies that can strengthen its structural heart leadership. Because these investments are typically smaller and focused on highly relevant areas, they can provide attractive long term optionality without creating excessive risk.
Unlock Exclusive Seeking Alpha Discounts – Level Up Your Investing With Zero Risk
If you’ve been thinking about improving your investing process, this is the easiest way to start. These offers are only available through my links, and the Premium plan even comes with a 100% risk-free 7-day trial. Try everything for a week, and if it’s not for you, just cancel. You lose nothing.
1) Seeking Alpha Premium — Try It Free for 7 Days
Access the tools I personally use every day:
• Earnings transcripts
• Stock screeners
• Deep-dive analysis
• Portfolio tracking
• Market news with context that actually matters
Special Price: $269/year (normally $299) + 7-day free trial (for new users only)
Try Premium Free for 7 Days → HERE
(Explore everything — cancel anytime during the trial and pay $0.)
2) Alpha Picks — Proven Stock Ideas
This stock-picking service has delivered +287% returns vs. the S&P 500’s +77% (July 2022–Nov 2025).Great for investors who want curated, long-term picks backed by data.
Special Price: $449/year (normally $499)
Get Alpha Picks → HERE
(Although Alpha Picks doesn’t offer a free trial, its historical outperformance means the subscription can often pay for itself quickly if results persist. For many investors, the potential return far outweighs the upfront cost).
3) Premium + Alpha Picks Bundle — Best Value
Get both services together and save $159.Perfect if you want both broad tools and high-conviction stock ideas.
Special Price: $639/year (normally $798)
Get the Bundle → HERE
(This bundle doesn’t include a free trial, but it gives you both services at a $159 discount. You get Premium’s in-depth research plus Alpha Picks’ high-performing recommendations, making it the most comprehensive option for serious investors.)
Valuation
Now it is time to calculate the share price. I perform three different calculations that I learned at a Phil Town seminar. If you want to make the calculations yourself for this or other stocks, you can do so through the tools page on my website, where you have access to all three calculators for free.
The first is called the Margin of Safety price, which is calculated based on earnings per share (EPS), estimated future EPS growth, and estimated future price-to-earnings ratio (P/E). The minimum acceptable rate of return is 15%. I chose to use an EPS of 1,81, which is from 2025. I have selected a projected future EPS growth rate of 12%. Finbox expects EPS to grow by 11,9% a year in the next five years. Additionally, I have selected a projected future P/E ratio of 24, which is twice the growth rate. This decision is based on the Edwards Lifesciences' historically higher price-to-earnings (P/E) ratio. Finally, our minimum acceptable rate of return has already been established at 15%. After performing the calculations, we determined the sticker price (also known as fair value or intrinsic value) to be $33,35. We want to have a margin of safety of 50%, so we will divide it by 2. This means that we want to buy Edwards Lifesciences at a price of $16,67 (or lower, obviously) if we use the Margin of Safety price.
The second calculation is known as the Ten Cap price. The rate of return that a company owner (or stockholder) receives on the purchase price of the company essentially represents its return on investment. The minimum annual return should be at least 10%, which I calculate as follows: The operating cash flow last year was 1.595, and capital expenditures were 260. I attempted to analyze their annual report to calculate the percentage of capital expenditures allocated to maintenance. I couldn't find it, but as a rule of thumb, you can expect that 70% of the capital expenditures will be allocated to maintenance purposes. This means that we will use 182 in our calculations. The tax provision was 217. We have 580,3 outstanding shares. Hence, the calculation will be as follows: (1.595 – 182 + 217) / 580,3 x 10 = $28,09 in Ten Cap price.
The final calculation is called the Payback Time price. It is a calculation based on the free cash flow per share. With Edwards Lifesciences' Free Cash Flow Per Share at $2,30 and a growth rate of 12%, if you want to recoup your investment in 8 years, the Payback Time price is $31,68.
Conclusion
I believe that Edwards Lifesciences is an intriguing company with strong leadership. It has built its moat through its extensive clinical evidence and physician trust, high switching costs, regulatory and manufacturing barriers, deep specialization in structural heart, and long standing relationships with hospitals and clinicians. The company has consistently achieved a high ROIC, and while it has declined over the past couple of years, it is expected to improve moving forward as newer growth areas mature and contribute more meaningfully to earnings. Edwards Lifesciences has also just delivered its highest free cash flow ever, and while free cash flow may remain somewhat volatile from year to year due to investments and one time factors, it is expected to grow over the long term. Competition is a risk for Edwards Lifesciences because the structural heart market is highly innovation driven and includes strong competitors such as Medtronic and Abbott Laboratories. If competitors introduce therapies with better outcomes, easier procedures, stronger durability, or lower cost, hospitals and physicians may gradually shift toward those alternatives, which could pressure both growth and margins. Laws and regulations are also a risk because the company operates in a highly regulated industry where delays in product approvals, changing rules, or compliance issues can directly slow product launches and affect growth. In addition, any safety issues or failure to comply with rules set by authorities such as the U.S. Food and Drug Administration can lead to fines, recalls, suspended sales, and reputational damage. Reliance on third party suppliers is another risk because the company depends on outside vendors for critical materials, components, manufacturing support, and sterilization, so any disruption can directly affect product availability and deliveries to hospitals. This risk is amplified by the use of single source suppliers and the lengthy regulatory process required to approve replacement vendors, which can lead to delays, missed sales, and potential reputational damage if product quality is affected. The transcatheter aortic valve replacement (TAVR) segment is a reason to invest in Edwards Lifesciences because it is the company’s largest business, built around the trusted SAPIEN platform, and supported by strong long term clinical data that reinforces physician confidence and expands patient access. In addition, newer evidence supporting earlier treatment and the focus on lifetime valve management create a long runway for durable growth over many years. The growing Transcatheter Mitral and Tricuspid Therapies (TMTT) segment is another reason to invest because it represents a second major long term growth engine beyond TAVR, with management targeting around $2 billion in revenue by 2030. Its broad portfolio of repair and replacement therapies, including PASCAL, EVOQUE, and SAPIEN M3, positions the company well to expand treatment options, deepen physician relationships, and drive revenue growth over the coming years. Innovation and expansion into new therapies are also reasons to invest because they provide multiple long term growth drivers beyond its core TAVR and TMTT businesses. Through next generation products, expansion into adjacent areas such as aortic regurgitation and heart failure, and disciplined acquisitions, the company continues to broaden its addressable market and strengthen its leadership in structural heart over many years. Overall, I believe there are many things to like about Edwards Lifesciences, and buying shares at the intrinsic value of the Ten Cap price at $56 could be a compelling long term investment.
My personal goal with investing is financial freedom. It also means that to obtain that, I do different things to build my wealth. If you have some extra hours to spare each month, you can turn a few hours a week into a substantial amount of money in a few years. If you are interested to know how I do it, you can read this post.
I hope you enjoyed my analysis! While I can’t post about every company I analyze, you can stay updated on my trades by following me on Twitter. I share real-time updates whenever I buy or sell, so if you’re making your own investment decisions, be sure to follow along!
Some of the greatest investors in the world believe in karma, and to receive, you will have to give (Warren Buffett and Mohnish Pabrai are great examples). If you appreciated my analysis and want to get some good karma, I would kindly ask you to donate a bit to 10.000 dogs. They rescue street dogs in Thailand by giving them food, medicine and vet care. If you have a little to spare, please donate here. Even a little will make a huge difference to save these wonderful animals. Thank you.




Comments